
Health Control
Anvisa updates rules and regulations for health measures in ports and airports
Due to the Ebola outbreak in Congo, preventive monitoring protocols were implemented in Brazil
Registration
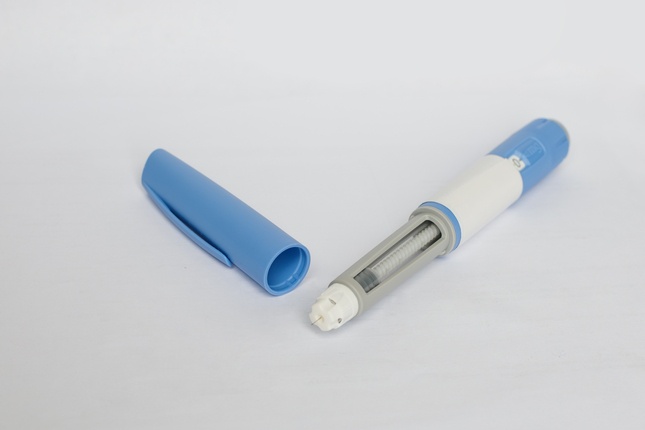
Anvisa approves the first synthetic semaglutide pen for diabetes, an analog product of Ozempic
The new product Ozivy underwent a rigorous technical evaluation of efficacy, safety and quality.

INTERNATIONAL AFFAIRS
Anvisa takes part in the 79th World Health Assembly on health regulation and global health
WHO’s initiative takes place in Switzerland and Brazilian delegation participates in debates on equity, access to health, pandemics, artificial intelligence and antimicrobial resistance
Joint Inspections
Anvisa and the Federal Police will conduct a joint analysis of weight-loss pens
Results will help investigations to assess health risks posed by irregular injectable GLP-1 medications
International Affairs
Anvisa receives representatives from US business entities
Meeting brought together members of the U.S. Chamber of Commerce and the Brazil-U.S. Business Council
Cooperation
Anvisa takes part in Portugal-Brazil Seminar on Regulatory Convergence
Regulatory authorities from both countries reaffirmed their commitment to strengthen technical cooperation.
Services
The Brazilian Health Regulatory Agency (Anvisa) is an autarchy linked to the Ministry of Health, part of the Brazilian National Health System (SUS) as the coordinator of the Brazilian Health Regulatory System (SNVS), present throughout the national territory.
Anvisa’s role is to promote the protection of the population’s health by executing sanitary control of production, marketing, and use of products and services subject to health regulation, including related environments, processes, ingredients, and technologies, as well as the control in ports, airports, and borders.



